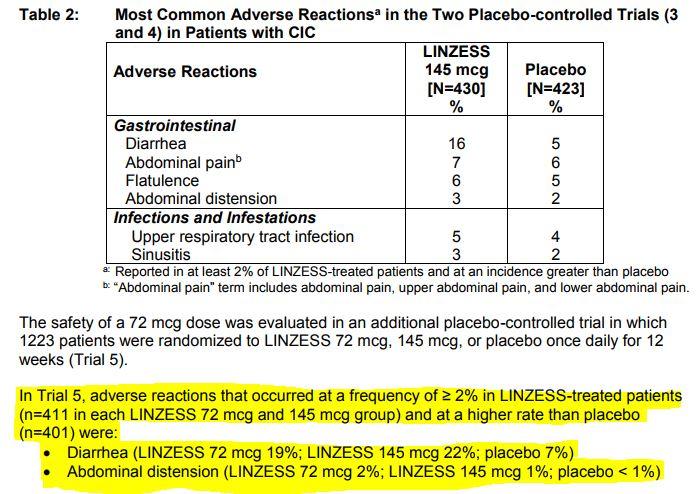
for the treatment of adult patients with irritable bowel syndrome with constipation (IBS-C) or chronic idiopathic constipation (CIC), based on IQVIA data. LINZESS® is the #1 prescribed brand in the U.S. We routinely post information that may be important to investors on our website at In addition, follow us on Twitter and on LinkedIn. 
We keep patients at the heart of our R&D and commercialization efforts to reduce the burden of GI diseases and address significant unmet needs.įounded in 1998, Ironwood Pharmaceuticals is headquartered in Boston, Massachusetts. Under the guidance of our seasoned industry leaders, we continue to build upon our history of GI innovation and challenge what has been done before to shape what the future holds. branded prescription market leader for adults with irritable bowel syndrome with constipation (IBS-C) or chronic idiopathic constipation (CIC). We are pioneers in the development of LINZESS® (linaclotide), the U.S. Ironwood Pharmaceuticals (Nasdaq: IRWD) is a leading gastrointestinal (GI) healthcare company on a mission to advance the treatment of GI diseases and redefine the standard of care for GI patients. "With this license agreement, we have now preserved the majority of LINZESS patent coverage for all three dosage strengths, reaffirming our commitment to grow the LINZESS franchise for many years to come," stated Tom McCourt, president and interim chief executive officer of Ironwood.Īs required by law, the companies will submit the settlement agreement to the U.S. Additional details regarding the settlements were not disclosed. District Court for the District of Delaware. That grant was in connection with a settlement resolving all previous Hatch-Waxman litigation between the companies and Teva regarding LINZESS patents pending in the U.S. Previously, Ironwood and Allergan granted Teva a license to market its 145 mcg and 290 mcg generic versions of LINZESS in the United States beginning on the same date. FDA approval), unless certain limited circumstances, customary for settlement agreements of this nature, occur. Pursuant to the terms of the agreement, Teva will be granted a license to market its 72 mcg generic version of LINZESS in the United States beginning Ma(subject to U.S. With this agreement, the companies have settled with the filers of all known ANDAs to date seeking approval to market generic versions of LINZESS. 
providing a license to Teva's abbreviated new drug application (ANDA) seeking approval to market a generic version of 72 mcg LINZESS® (linaclotide) prior to the expiration of the companies' applicable patents. (NASDAQ: IRWD), announced today that, with its partner AbbVie Inc., the companies have reached an agreement with Teva Pharmaceuticals USA, Inc. – The earliest licensed entry of any generic linaclotide 72 mcg, 145 mcg or 290 mcg in the U.S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
